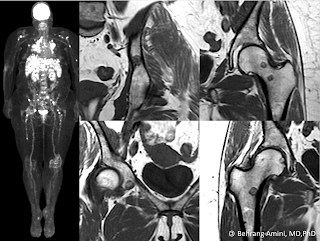
Neurofibromatosis type 1
Neurofibromatosis type 1 (NF-1), in addition to a neuroectodermal disorder, is accompanied by mesodermal dysplasia that is accompanied by skeletal changes. The typical osseous findings include
bowing of the legs, increase in length of long bones, pseudarthrosis, subperiosteal cyst formation, local bony erosions from adjacent lesions, and intramedullary neurofibromas. Except for the last two, which are due to direct involvement by neurofibromas, the remainder are due to dysplastic changes in bones.
A lesser known osseous presentation in bone is the propensity for subperiosteal hemorrhage and hematoma formation. The cause is unknown, but may be related to:
- Vascular abnormalities: For example, diffuse flat hemangiomas or plexiform dilated veins, which have been described in patients with hypertrophy of the extremities
- Dysplastic periosteum: The thinking is that mesodermal dysplasia manifests as an abnormally loose periosteum with poor callus response. This would predispose the patient to the formation and propagation of large subperiosteal hematomas.
- Direct involvement by neurofibromas: Subperiosteal infiltration by neurofibromatous tissues may loosen the periosteum and allow for massive hemorrhage following minor trauma.
References